

 更多“Lack of Zinc _____causes a ran…”相关的问题
更多“Lack of Zinc _____causes a ran…”相关的问题
ong as the concentration of L excess that of zinc (II) by a factor of 5, the absorbance of the solution is only lined on the cation concentration. Neither zinc(I) nor L absorbs at 600 nm. A solution that is 1.60X10-6mol·L-1in zinc (II) 1.00 mol·L-1in L has an absorbance of
0.164 in a 1.00 cm cell at 600 nm.Calculate
(1) the transmittance of this solution.
(2) the transmittance of this solution in a 3 cm cell.
(3) the molar absorbance of the complex at 600 nm.
A.expand
B.extent
C.extend
D.expire
A.short of
B.missing
C.lack in
D.charging
家族性矮身材
A.HA=BA<CA(CA:时间年龄,BA:骨龄,HA:身高年龄)
B.HA<BA—CA
C.BA<HA<CA
D.BA>HA>CA
E.HA=BA=CA
甲状腺功能低下
A.HA=BA<CA(CA:时间年龄,BA:骨龄,HA:身高年龄)
B.HA<BA=CA
C.BA<HA<CA
D.BA>HA>CA
E.HA=BA=CA
A.HA=BA<CA
B.BA<CA<HA
C.BA<HA<CA
D.HA<BA=CA
E.HA=BA>CA
A.体质性生长迟缓:HA=BA B.家族性矮身材:HA C.甲状腺功能低下:BA D.垂体功能减低:HA E.以上都正确
已知298K时,Ca(OH)2(s)的 =-897.5kJ·mol-1,H2O(1)的
=-897.5kJ·mol-1,H2O(1)的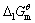 =-237.1kJ·mol-1,
=-237.1kJ·mol-1,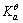 = 1.00×10-7,试求电对Ca(OH)2/Ca的电极电势。
= 1.00×10-7,试求电对Ca(OH)2/Ca的电极电势。